AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
Back to Blog
Maleic acid sigma and pi bonds9/12/2023  
In human beings, when we shake hands with someone, we can hold out one arm and shake hands with them without any difficulty. The atom puts its hand out to its partner and makes a single bond. So how does a carbon atom bond with another atom or molecule in a single bond (bond to one place)? Naturally, the atom chooses the easiest way. 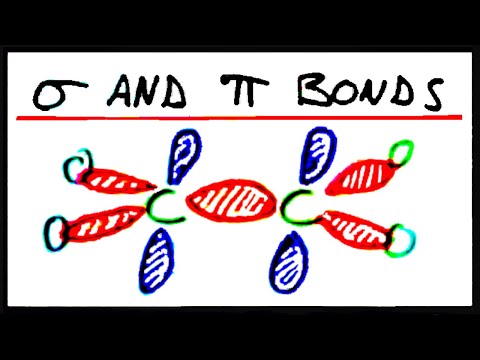
σ-Bond (Sigma Bond) Forms a Covalent Bond and the Bond Energy Is HighĬarbon atoms can bond with other atoms and molecules using their four hands. So at first, let’s just understand that a carbon atom has four hands, and can bond with other atoms and molecules. However, it becomes complicated with words like s orbital, p orbital, and sp3 hybridization. Therefore, each hand repels the other, resulting in each hand pointing in a different direction. The four hands are not free to move, and they are pointing in different directions.Įlectrons have a negative charge. In the case of sp3 hybrid orbitals, there are hands in various directions. It should be understood that there are four hands in a carbon atom if there are four hands due to an s orbital and p orbital, this is called on sp3 hybridization. In ethane, one carbon atom is bonded to four other atoms. In other words, a carbon atom can be bonded in four places. If we focus on the single-bonded carbon atom, the carbon atom has one s-orbital and three p-orbitals, which means that there are four hands. These orbitals are called s orbital and p orbital. An atom’s hand refers to its electron orbitals.Įach atom or molecule has orbitals. When atoms bond, they need to give their own hand. The sp3 Hybrid Orbitals Have Four Hands in s and p Orbitals There are two types of bonds between molecules: σ-bond and π-bond, and we must clearly distinguish between σ-bond and π-bond. When molecules are bonded, they are not bonded in this simple way, but actually bonded in a special way. As long as you have this image in your mind, you will not be able to understand the sigma and pi bonds in chemistry. What image do you have in mind when you understand the double bond of a compound? The majority of people who have taught high school chemistry, including when assembling a model of a molecule, have the following image of a bond.įorget about this molecular image. We Should Forget the Image of Double Bonds in High School Chemistry
0 Comments
Read More
Leave a Reply. |
 RSS Feed
RSS Feed

